How a Catalytic Converter Works: A Complete Guide
Understanding How a Catalytic Converter Works: Its Role In Reducing Pollution
The catalytic converter plays a crucial role in reducing harmful emissions produced by the engine. Despite its importance, many drivers don’t understand what a catalytic converter does, how it works, or why it’s so vital for both vehicle performance and environmental protection. This article will explore the catalytic converter’s function, the science behind it, and its significance in the broader context of automotive engineering and environmental regulation.
A catalytic converter is part of the exhaust/emissions system
A catalytic converter is part of the exhaust system and is usually mounted right after the exhaust manifold. It contains a ceramic honeycomb coated with a microscopic layer of precious metals like Platinum, Palladium, Rhodium, and Cerium, along with iron, manganese, and nickel. The exact mix of metals depends on the engine and the current state of emissions laws.
The metals were chosen because they can store excess oxygen in the exhaust stream. Then, they combine that stored with unburned hydrocarbons to clean up the exhaust and reduce emissions. The precious metals also react with oxides of nitrogen and turn them into water and nitrogen.
Think of a catalytic converter like an incinerator whose job it is to burn off excess gasoline, oil, and coolant that gets into the exhaust. So, it reduces emissions of hydrocarbons, oxides of nitrogen, and carbon monoxide.
What does a catalytic converter do, and how does it work?
The first generation of converters was referred to as 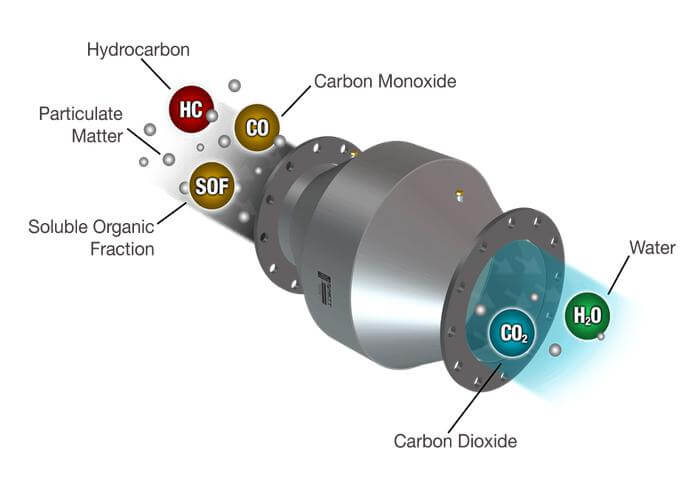 two-way converters because it oxidized carbon monoxide, giving it an extra oxygen atom—2CO + O2 → 2CO2. It also oxidized unburned hydrocarbons (gasoline and oil), turning them into carbon dioxide and water.
two-way converters because it oxidized carbon monoxide, giving it an extra oxygen atom—2CO + O2 → 2CO2. It also oxidized unburned hydrocarbons (gasoline and oil), turning them into carbon dioxide and water.
However, two-way converters couldn’t eliminate oxides of nitrogen, the main component of smog. Oxides of nitrogen are considered a greenhouse gas and contribute to acid rain. In addition, they’re an ozone-depleting substance 300 times more potent than carbon dioxide (according to Wiki). So, carmakers switched to three-way catalytic converters starting in 1981 to reduce oxides of nitrogen emissions.
A three-way catalytic converter converts oxides of nitrogen to nitrogen and oxygen in addition to the two jobs performed by the previous two-way converters.
How a catalytic converter works: The chemistry behind the process
The catalytic converter relies on several key chemical reactions to achieve its emission-reducing effects:
How a Catalytic Converter Works to Reduce Nitrogen Oxides (NOx):
Nitrogen oxides are reduced to nitrogen and oxygen, which are harmless gases naturally present in the atmosphere.
2NOx→xO2+N2
2NOx→xO2+N2
How a Catalytic Converter Oxidizes Carbon Monoxide (CO):
Here’s how a catalytic converter works to convert Carbon monoxide to carbon dioxide, a less harmful gas, although still a contributor to greenhouse gases.
2CO+O2→2CO2
2CO+O2→2CO2
How a Catalytic Converter Oxidizes Hydrocarbons (HC):
Here’s how a catalytic converter works to get rid of excess hydrocarbons in the exhaust stream. The converter oxidizes the HC into carbon dioxide and water vapor, both of which are significantly less harmful to the environment.
CxHy+(x+y)4O2→xCO2+y2H2O
CxHy+4(x+y)O2→xCO2+2yH2O
These reactions occur simultaneously as the exhaust gases flow through the catalytic converter, effectively reducing the levels of harmful pollutants emitted from the vehicle’s tailpipe.
How long does a catalytic converter last
There are no wear parts inside a catalytic converter, and its oxygen-storing and pollution-reducing capabilities never wear out on their own. However, it can be damaged by being fed too much motor oil, gasoline, coolant, or silicone sealants.
A catalytic converter can last the life of the vehicle if you don’t abuse it. Catalytic converters don’t die on their own—they’re murdered. Here’s the bottom line: Catalytic converters do NOT wear out; they are killed by the owner’s negligence. That’s right, YOU kill your catalytic converter by ignoring misfires, not fixing oil and coolant leaks, and by hitting things with the undercarriage.
What causes a Catalytic Converter to Fail?
An engine misfire is the number one cause of catalytic converter failure. When a cylinder misfires, it dumps raw fuel into the converter, causing it to overheat as it oxidizes the excess fuel. If the misfire continues long enough, the converter can overheat to beyond 1,300 degrees, causing the ceramic honeycomb to melt.
That plugs up the openings in the honeycomb, restricting exhaust flow and reducing the engine’s ability to breathe. A meltdown can also cause the honeycomb “brick” to shatter, causing a catalytic converter rattle sound. A converter can also be damaged by impact or contamination from a leaking cylinder head gasket that sends coolant into the exhaust.
©, 2015 Rick Muscoplat
Posted on by Rick Muscoplat