Recondition a Car Battery: Why It Doesn’t Actually Work
Why Reconditioning a Car Battery Doesn’t Work
Quick Summary
Reconditioning a car battery rarely works. Once a battery has developed hard sulfation or suffered plate damage from acid stratification or freezing, it is physically impossible to restore it. Despite what online videos and chemical “hacks” claim, most attempts to recondition a car battery don’t fix the underlying damage—and often leave you stranded shortly afterward.
Article
Why Reconditioning a Car Battery Doesn’t Really Work
Car batteries usually don’t fail suddenly or mysteriously. They wear out in very predictable ways, and once that damage crosses a certain point, no chemical trick or charger can undo it. I’ve tested hundreds of batteries over the years, and every time someone asks whether they can recondition a car battery, the answer comes down to understanding what’s happening inside the case.
As a lead-acid battery discharges and sits unused, two destructive processes begin almost immediately. If those processes progress far enough, the battery is done—no matter what the internet says.
What Actually Happens Inside a Failing Car Battery
The first problem is sulfation. Any time a battery discharges, sulfate crystals form naturally on the lead plates. If the battery is recharged quickly, those crystals dissolve back into the electrolyte. That part is normal. The trouble starts when the battery sits in a discharged or partially discharged state for too long.
Over time, those soft sulfate crystals harden into a dense, non-conductive layer. Once that happens, internal resistance skyrockets. At that point, it becomes impossible to force enough current through the battery to recharge it properly. When people ask me if they can recondition a car battery, this is usually the failure mode they’re up against—and hard sulfation cannot be reversed.
The second issue is acid stratification. A fully charged car battery contains roughly a 63% water and 37% sulfuric acid mixture. When a battery sits unused, the heavier sulfuric acid sinks to the bottom of the case while lighter water rises to the top. This creates a highly concentrated acid layer at the bottom that aggressively eats away at the plates.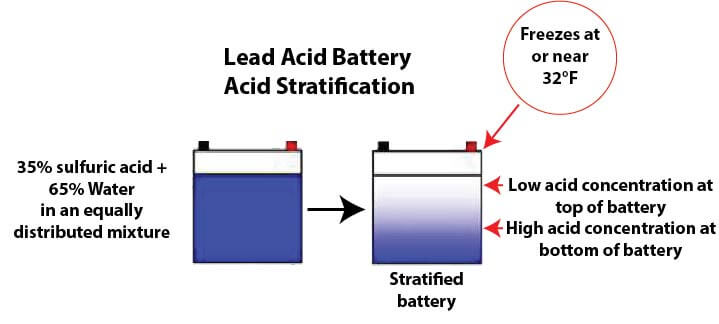
Once plate material is physically damaged or eroded, there is no process—chemical or electrical—that can rebuild it. None. This alone makes it impossible to truly recondition a car battery that has been sitting dead for an extended period.
If you’ve tried reconditioning the battery with a desulfating charger and the battery still fails a resistance, capacitance, and load test, there is no way to revive it. None. The battery is dead, and it will stay dead.
Why Chargers and “Desulfators” Rarely Save a Dead Battery
Modern smart chargers with desulfation modes can help only in very early stages of battery sulfation. If a battery still passes resistance, capacitance, and load tests after charging, it may have some life left. But if it fails those tests, no charger can change that outcome.
Once a battery fails under load, there is no safe or reliable way to recondition a car battery back to full service. Anyone telling you otherwise is ignoring basic electrochemistry.
Why Chemical Battery Reconditioning Hacks Don’t Work
The internet is full of advice claiming you can recondition a car battery by adding household chemicals. I’ve seen every version of these hacks, and none of them repairs the actual damage.
One popular suggestion is adding distilled water to revive a dead battery. If the electrolyte level is already above the plates, low water isn’t the cause of failure. Adding water won’t restore lost capacity or reduce internal resistance. If the plates are already exposed, the battery is permanently damaged—and adding water won’t change that.
Another common method involves draining the battery, neutralizing the acid with baking soda, and then refilling it with an Epsom salt solution. Yes, this may generate enough voltage to start an engine once or twice. But it doesn’t reverse sulfation or plate erosion. It’s not reconditioning—it’s a short-lived chemical illusion.
The same goes for crushed aspirin mixed with water. Aspirin is an acid. Adding more acid may temporarily spike voltage, but it doesn’t rebuild damaged plates. This approach does not recondition a car battery in any meaningful or lasting way.
When Battery Reconditioning Appears to “Work”
Here’s the uncomfortable truth: when people say they successfully reconditioned a car battery, the battery usually wasn’t dead to begin with. It was discharged, neglected, or mildly sulfated—but not structurally damaged.
In those cases, a slow recharge or desulfation cycle didn’t recondition anything. It simply returned the battery to the condition it should have been in all along.
That’s a critical distinction most online guides never mention.
My Professional Verdict on Battery Reconditioning
If a battery cannot fully recharge and pass proper electrical testing, it cannot be revived. Period. No chemicals, no shaking, no secret formulas. When people ask me whether it’s worth trying to recondition a car battery, my advice is simple: test first, believe the results, and don’t rely on internet myths.
Replacing a failing battery before it leaves you stranded is cheaper than towing, missed appointments, and electrical system stress caused by low voltage.
©, 2020 Rick Muscoplat
Posted on by Rick Muscoplat