Understanding Battery Sulfation: Causes and Solutions
Understanding Battery Sulfation and its Causes
Sulfation occurs when lead sulfate crystals form on the battery’s lead plates, impairing its ability to hold and deliver a charge. This process can significantly reduce the lifespan and efficiency of a battery. Understanding the causes of sulfation and how to prevent it is crucial for maintaining battery health and performance.
Several factors can contribute to the formation of lead sulfate crystals and the onset of sulfation:
• Undercharging— One of the most common causes of sulfation is undercharging, where the battery is not charged fully or is left in a discharged state for extended periods. When a battery is not fully charged, the lead sulfate formed during discharge is not completely converted back, leading to the gradual accumulation of sulfate crystals.
• Deep Discharge— Deeply discharging a battery, especially repeatedly, can cause severe sulfation. When a battery is discharged beyond a certain point, lead sulfate crystals can form more rapidly, and these crystals may become difficult to dissolve during recharging.
• Long Storage Periods— Storing a battery for long periods without use can also lead to sulfation. In a discharged state, lead sulfate crystals can form and harden over time, even without a load on the battery. This is particularly problematic in environments with high temperatures, which accelerate the sulfation process.
• Low Electrolyte Levels— In lead-acid batteries, the electrolyte level should be maintained above the plates to ensure proper chemical reactions. If the electrolyte level drops due to evaporation or leakage, the exposed plates can sulfate, as they are no longer fully immersed in the acid solution.
• Temperature Extremes— Extreme temperatures, both high and low, can affect the chemical reactions within a battery. High temperatures accelerate the rate of chemical reactions and can increase the rate of sulfation, while low temperatures can slow down the conversion of lead sulfate back into active materials during recharging.
How does a battery sulfation happen?
This is a simplified explanation. Batteries produce power through a chemical reaction between the lead plates and the electrolyte (battery acid). Car battery acid is a diluted solution of sulfuric acid H2SO4. In simple terms, the battery acid provides the sulfate, hydrogen, and oxygen needed to interact with the pates and produce power.
When a car battery is discharged, the sulfate in the acid is attached to the surface of the plates. The sulfate crystals reduce the plate’s surface area, which, in turn, reduces the battery’s ability to produce power. Worse yet, the sulfate crystals act as an electrical insulator, increasing the battery’s internal resistance (keep this in mind when we cover recharging). At this point, the battery acid has given up most of its supply of sulfate and hydrogen. All that’s left is water H2O. That’s why discharged car batteries freeze in cold weather.
Recharging reverses the sulfation process — to a point
When the alternator is operating properly, it literally pushes the sulfate and hydrogen off the plates and forces it back into solution, which turns the H2O back into H2SO4. But it doesn’t always work that way, as you’ll see below.
Two types of battery sulfation
There are two kinds of car battery sulfation; the reversible (soft crystal) type and the permanent (hard crystal) type. If reversible sulfation is caught early, it can be eliminated by recharging at the proper rate. However, if the sulfation is ignored and left in place for weeks or months, large portions of the lead plate surface area will become permanently unavailable. That dramatically reduces the battery’s ability to produce starting power. It may fail to produce enough power to operate the starter in warm weather and may even fail to provide enough power to light the lights in cold weather.
The longer a battery sits in a discharged state, the more likely the battery is permanently damaged. As a battery sits in the discharged state, the sulfate crystals grow in size and harden. After a certain period of time, they’re impossible to remove, even with recharging. At that point the battery must be replaced.
Can you desulfate a battery?
You can desulfate a battery if the sulfation is the reversible soft crystal type. But the recharging process is somewhat tricky. For example, a battery charger that’s equipped with a six-phase battery reconditioning routine may start by recharging process at a low voltage, around 4 volts and 7 amps. That’s called the energizing phase. Why low voltage? Because a sulfated battery has developed high internal resistance. If you hit it with high voltage right off the bat, that high resistance will create HIGH HEAT, and that heat will warp the plates and short out the battery.
Next, the charger boosts the charging current to around 13.5 amps while slowly ramping up the voltage to a peak of around 14.5 in phase 3. In the third or absorption phase, it maintains constant voltage while ramping down the amps. The 4th phase drops the voltage slightly and the amperage a bit further. In the 5th or resting phase, it drops the current flow to 0 while maintaining around 12.5 volts. The 6th phase exercises the battery by pulling a load on the battery until it reaches the final restoration phase, where it raises amperage and voltage one last time. The key to reconditioning a battery after sulfation is to prevent overcharging and overheating.
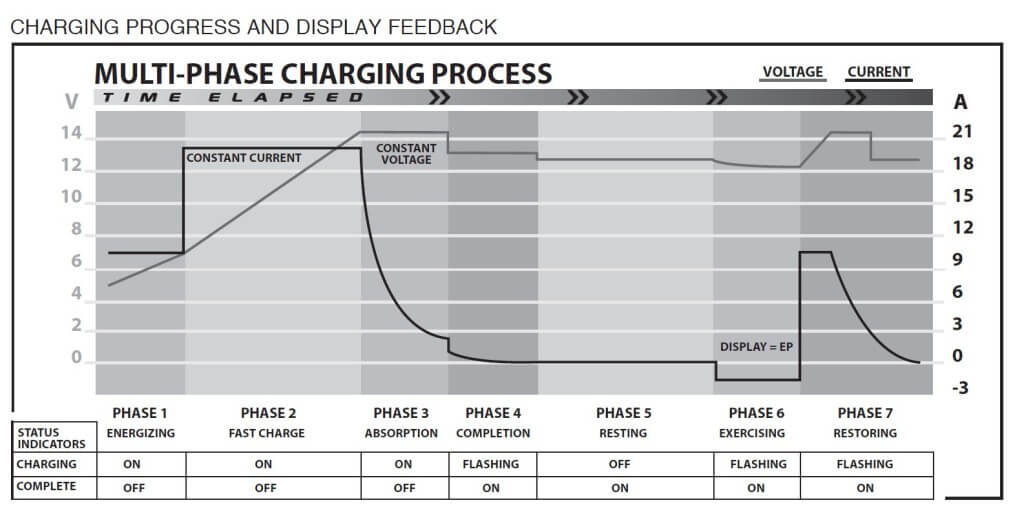
This chart shows the six phases of battery reconditioning — Clore Automotive
What about those “miracle” battery desulfation chargers?
Some companies promote a battery-desulfating charger that works by introducing a range of high-frequency pulses at low voltage. The pulses supposedly break up and dissolve the sulfate crystals in a short time period. Do those devices work? Partially. They can reduce soft crystal sulfation, But I’m not convinced they reduce large hard sulfate crystals. s
Here’s a comment from BatteryUniversity.com on the effectiveness of pulse desulfation:
“Applying random pulses or blindly inducing an overcharge can harm the battery by promoting grid corrosion. There are no simple methods to measure sulfation, nor are commercial chargers available that apply a calculated overcharge to dissolve the crystals. As with medicine, the most effective remedy is to apply a corrective service for the time needed and not longer.
While anti-sulfation devices can reverse the condition, some battery manufacturers do not recommend the treatment as it tends to create soft shorts that may increase self-discharge. Furthermore, the pulses contain ripple voltage that causes some heating of the battery. Battery manufacturers specify the allowable ripple when charging lead acid batteries.”
How to prevent car battery sulfation?
• Regular Charging— To prevent undercharging, it is essential to charge the battery regularly and fully. For vehicles or equipment that are not used frequently, a maintenance charger can be used to keep the battery charged and prevent sulfate buildup. Don’t just start your car in cold weather and let it idle. The alternator doesn’t provide much power at idle speeds. DRIVE IT on the highway to get the RPMs up.
• Avoid Deep Discharges—Avoiding deep discharges is crucial for prolonging battery life. To minimize the risk of sulfation, keep the battery’s charge level above 50%. Many modern devices come with battery management systems that prevent deep discharges.
• Proper Storage— When storing batteries for extended periods, it is important to store them fully charged and in a cool, dry place, or connect them to a battery maintainer. Checking the charge periodically and recharging if necessary can prevent sulfation during storage.
• Maintaining Electrolyte Levels— Regularly check the electrolyte levels in flooded lead-acid batteries and top up with distilled water if needed. Keeping the plates submerged in the electrolyte is essential to prevent exposure and sulfation.
• Temperature Control— Avoid exposing batteries to extreme temperatures. In hot climates, ensure proper ventilation and avoid direct sunlight. In cold climates, use battery heaters or insulation to maintain a suitable temperature.
• Maintain your charging system— Make sure the alternator drive belt and the belt tensioner are in good condition to prevent slipping. See this post on checking your serpentine belt and belt tensioner.
• Clean your battery terminals— Corroded terminals create high resistance and that reduces the effectiveness of your alternator.
©, 2019 Rick Muscoplat
Posted on by Rick Muscoplat